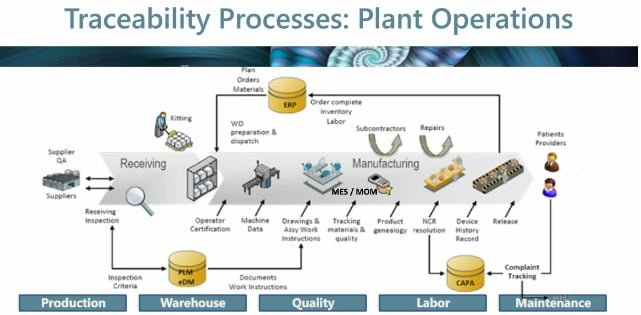
Medical device developers are evolving and introducing new products and variants into the market. With this evolution comes a more complex and dynamic global supply chain. To help navigate this shifting terrain Dassault Systèmes generated the free webinar below to help guide developers through effective techniques to create an opportunity through traceability.
Check out the 35 minute free Qmed webinar on Making Traceability Work for the Business: Effective eDHR and UDI to discover:
- The role of integrated manufacturing software in eDHR and UDI compliance
- Approaches to consider for traceability program improvements
- Questions to raise as you assemble the diverse stakeholders for such a project