The American Medical Device Summit is less than one month away! The summit will draw on the knowledge of over 40 industry leaders to explore the challenges and opportunities in product development, quality management, speed to commercialization, and regulatory harmonization.
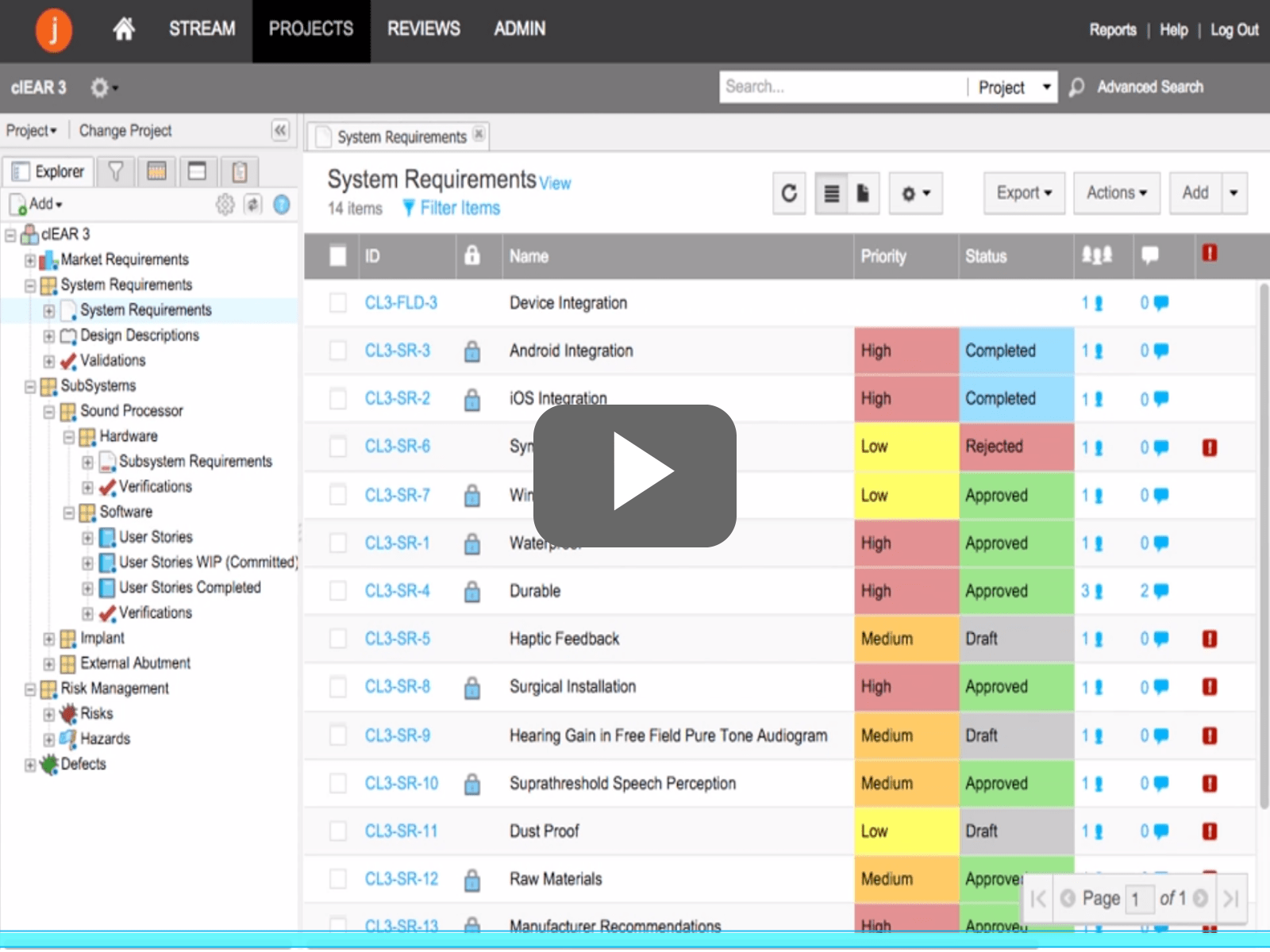
One of the companies helping developers tackle the challenges in product development and compliance is Jama Software. Jama Software has developed a more agile alternative to document-centric methods for requirements management, creating the optimal environment for defining problem statements, validating cross-functional input and ensuring that what is delivered at each phase of is what was intended.
Check out the Demo Video below to find out how on-boarding this software can help developers navigate complex development cycles and build great products by:
- Removing the overhead of requirements reviews.
- Shortening the time between milestones.
- Mitigating risk and maintaining traceability evidence.
- Visualizing connections between regulations, requirements and test cases.
for a more in-depth look at the software, try the software yourself by signing up for the Jama Software Free Trial!